
Olga Schäfer
Johannes Gutenberg University Mainz, Germany
Title: Secondary structure-driven self-assembly of reactive polypept(o)ides: Controlling size, shape und function of core-crosslinked nanostructures
Biography
Biography: Olga Schäfer
Abstract
The rational development of nano-sized delivery systems remains a challenge in the materials as well as bio-medical sciences, especially when independent control over size, shape and functionality of the carrier is desired. Herein, we report on nanostructures derived from amphiphilic block copolypept(o)ides1, based entirely on endogenous amino acids. Controlled self-assembly allows for a strategy to adjust core polarity separately from particle preparation in a bio-reversible fashion. Additionally, the peptide-inherent process of secondary structure-directed self-assembly allows for morphology control of core cross-linked nanostructures from the same polymer precursor, offering a simple yet powerful approach to versatile peptide-based nanoparticles for delivery of various therapeutic relevant agens such as chemotherapeutic drugs or siRNA2.
A recently developed S-ethylsulfonyl protective group3,4 enables controlled polymerization of α-amino acid N-carboxyanhydrides (NCAs), directs self-assemblyin water and allows core-crosslinking by a fast chemoselective reaction into bio-reversible disulfides. The hydrophobic block is further able to form β-sheets,5leading to unidirectional aggregation in aqueous solution and ultimately to worm-like particles, or, after suppression of secondary structures, to spherical micelles. Independent from the morphology, the crosslinker dictates the functionality of the nanoparticle core and enables the introduction of cationic moieties for siRNA complexation. Given the bioreversible nature of disulfide bonds, they respond to differences in reduction potential and hence provide stability in extracellular medium, while they are cleaved inside the cell and release the cargo.
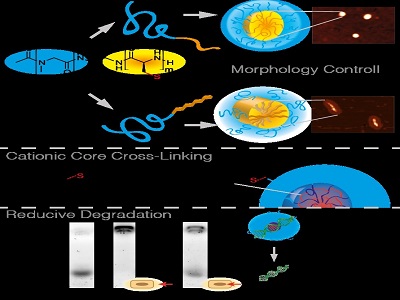
Figure 1: Self-assembly of amphiphilic PSar-b-PCys(SO2Et) block copolypept(o)ides yields spherical or rod-like nanoparticles, depending on the secondary structure of the polymer. After self assembly the core functionality is introduced by disulfide core-crosslinking, which proves to be bioreversible.

